Using TENGs to Build a Implantable Self-Charging Pacemaker
| 18-08-2021 | By Liam Critchley
Self-powered implantable devices are thought to be the next generation of technologies in the medical device sector (and in medicine/surgery in general). They not only have the potential to operate over much longer timescales, but they also reduce the need for repeated surgery (that is currently required to charge the device after a certain period). Implantable medical devices that can monitor the internal environments and modulate any abnormalities are very sophisticated. Care needs to be taken with the choice of materials and how the devices function.
Many of the implantable devices used today are not self-charging and face some key challenges that contribute to a higher long-term financial burden and a higher risk to patient health. This is because non-self-powering devices have finite lifetimes to be used, so multiple surgeries are needed to repair and replace the device once it has reached its usable life. However, the ability to self-charge can negate the need for numerous surgeries by powering the device from its surroundings and removing the need to remove the device because it has run out of power (only if there are medical issues/malfunctions).
There are a number of energy harvesting devices out there nowadays, including many that are very small. However, we have not seen many implantable medical devices becoming self-charging in a clinical capacity. There is currently a lack of in-vivo harvesting devices that can produce a sufficient charge to power complex medical devices such as pacemakers. While medical devices are in trial with different in-vivo energy harvesters, new avenues are constantly being sought. One of the latest areas to be trialled is triboelectric nanogenerators (TENGs).
Moving Away from MEMS for Implantable Medical Devices
While there are now many self-powered medical devices for diagnostic and monitoring applications outside of the body (e.g., skin sensors and wearable electronics), the body's internal environment makes self-powering inherently more challenging for complex devices. In this field, there has been a lot of interest in creating devices that use microelectromechanical systems (MEMS)—driven by elastic motion—to power themselves.
A lot of MEMS are using spring suspensions to generate power from human motions. However, in in-vivo environments, these systems are not as effective because the movement of the human body is not continuous (and needs to be for these systems to work), and for pacemakers specifically, many of these MEMS systems do not produce enough power to charge them. So, while MEMS have been useful for charging many other devices, they are not suitable for pacemakers, so other in-vivo energy harvesting devices are also in a trial.
Triboelectric Nanogenerators (TENGs)
One of the latest approaches across a lot of in-vivo and external medical monitoring devices has been to use triboelectric nanogenerators (TENGs) to harvest motion from their surroundings. TENGs are also typically low-cost devices and are lightweight, so they could theoretically be suitable for clinical applications. A nanogenerator converts an external stimulus into an electrical current, which can then power the device. TENGs are the branch of nanogenerators that are involved with changing mechanical energy into electrical energy.
TENGs typically rely on two fundamental mechanisms to convert mechanical motion into an electrical output: contact electrification and electrostatic charge induction mechanisms. For things to work, they first become electrically charged from the interaction with an external stimulus. The contact electrification mechanism is responsible for this and generates frictional forces, which produces electrical charges on the surface of the TENG. The TENG then redistributes these charges across the materials within the nanogenerator, enabling a current to be generated that can power a small-scale device. This second phase is the electrostatic charge induction mechanism, and both mechanisms work harmoniously to power a device from its surroundings.
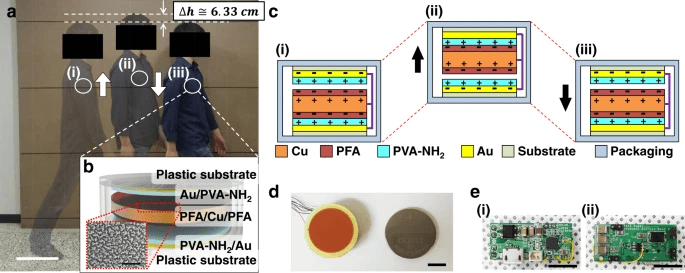
a Photograph of a walker showing a vertical displacement of ~6 cm. scale bar, 30 cm. b Schematic image of the I-TENG, and surface SEM image of the surface-modified PFA layer. scale bar, 5 μm. c The working mechanism of the I-TENG with the freestanding unit. d Photograph of the stacked I-TENG and a commercial coin battery. scale bar, 1 cm. e Photographs of the (i) front and (ii) back of the power management system. scale bar, 1 cm.
Creating a Self-charging Pacemaker with Inertia-Driven TENGs (I-TENGs)
Researchers from South Korea have created a high-performance inertia-driven triboelectric nanogenerator (I-TENG). The I-TENG is the size of a coin battery and functions using a combination of body motion and gravity. The nanogenerator itself was created by layering different materials on top of each other. The materials include copper and gold, alongside two different polymer-based triboelectric materials: amine-functionalised poly(vinyl alcohol) (PVA-NH2) and perfluoroalkoxy (PFA).
The development of the TENG and its integration into a pacemaker went through several stages. The team first demonstrated the potential of the I-TENG itself at the initial tests. It showed that it could produce a volume power density (for converting mechanical energy into electricity) of 4.9 μW/cm3 at a load resistance of ~10 MΩ, which is competitive with a lot of other high-performance energy harvesters. It was also found that the I-TENG could harvest z-axis mechanical energy and was not strongly influenced by x- and y-axis motion.
The researchers then moved onto a preclinical test using large animals, and the test involved inserting the I-TENG into different places of an animal host. The I-TENG data was collected and monitored in real-time via Bluetooth. The preclinical test showed that charging a Li-ion battery using the I-TENG in an in-vivo biological environment was possible.
After a successful first phase of the preclinical trial, the team inserted the I-TENG into a cardiac pacemaker in the second phase. In this preclinical trial, the I-TENG was able to charge the Li-ion battery in the pacemaker with the help of a power management integrated circuit (PMIC). Moreover, the I-TENG charged up capacitors while the animal was asleep.
In an area where there is a need for self-charging devices and few options for clinical use, this research opens a door for developing new self-rechargeable implantable medical devices. While the preclinical trials look promising, there is still a lot of work to go until pacemakers such as these end up being clinically approved. Still, it lays the foundations for a potential new route for realising self-charging implantable medical devices in the future.
Reference:

